MatDeck has a variety of time-saving and unique features that have been specially tailored to help the needs of any electrochemists. The field of electrochemistry itself concerns itself with the relationship between electrical potential as a quantifiable and quantitative phenomena and detectable chemical change, with electrical potential as an outcome of a specific chemical change. MatDeck can help chemist predict, create and study these electrochemical reactions, with features such as east to use DAQ control to create and sustain electrochemical reactions to unique and easy-to-use Formula Templates, which allow the user to solve equations within a few clicks and no writing needed.
MD Drag-and-Drop Formula in Electrochemistry
Formula Templates are ready-made equations, lines of code or any frequently used GUIs. MatDeck offers a variety of different Formula Templates for all subjects, including Chemistry, Physics, DSP, FFT and now Electrochemistry. The idea behind them is to take away any repetitive writing or mathematical calculation as well as eliminating all human error. This can be seen below in the Nernst equation and its derivatives.
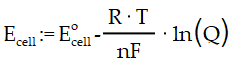


These formulas are somewhat lengthy, picky and require attention to detail to ensure you have all variables, powers and operators correctly. Instead, users can head over to the Insert tab and on the far right they can chose the Field of the Template they want to add.

Once you select the field, all you then need to choose the Formula from the dop down menu below.

Then all you need to do is Click the Insert button and place the Formula Template anywhere inside the document and now you can solve the equation by just changing the value of the variables. As you can see within a few click you have added and solved a complex equation. While this may seem like a inefficient solution with the simple formulae in the picture above, it is a life saver when using some more realistic electrochemistry formulae such as the MD Formula Templates below.




As you can see from the images above, equation such as the Tafel equation are key and incredibly intricate formulas, in MatDeck it is nothing more than a few clicks.
Electrochemistry Formula Templates – Faradays First Law of Electrolysis:
The mass of primary products generated by electrolysis at an electrode is proportional to the amount of electricity passed through it. According to the first law, the amount of chemical change caused by a current at the electrode-electrolyte interface is proportional to the amount of electricity consumed.

The Z is the proportionality constant which also called electro-chemical equivalent.

Electrochemistry Formula Templates – Faradays Second Law of Electrolysis:
The second states that the amounts of chemical changes caused by a given amount of electricity (m1,m2) in various substances are proportionate to their corresponding weights(W1,W2).

A Faraday is the amount of power required to create a chemical change in one equivalent weight unit (F).ξ is the stoichiometric factor, M is the molecular weight. F is the Faraday constant and Q is the total electric charge transferred to or from the electrode in coulomb.

Electrochemistry Formula Templates – Faradays Law and Corrosion Electrochemistry
Corrosion is an electrochemical process in which electrons are transported between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate in both aquatic and atmospheric environments.

N is the number of moles, n is the number of electrons per molecule of the species being reacted, I is the total current in amperes (A), t is the period of the electrochemical method in seconds. you were familiar with the current generated by one of the anodic reactions, you might use a useful relation developed by Michael Faraday to convert this current to a similar mass loss or corrosion penetration rate.

ΔN is the change in that quantity, n is the number of electrons per molecule of the species being reacted, I is the total current in amperes (A), t is the period of the electrochemical method in seconds (s).M/n is equivalent weight, RM is the corrosion rate, icorr is corrosion current, ρ is the density and F is Faraday constant.
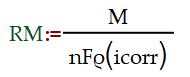
Electrochemistry Formula Templates – Coulomb’s Law Formula
In electrolyte solution, Coulomb’s law is used to explain the forces of attraction between charges.

We must make changes in above mention physics formula to be able to use it for oppositely charge ions. In Columbus law the constant k is written as.

Where the permittivity of free space, such as a vacuum, is ε0 However, because we’re dealing with a solution, we must take into account the effect of the medium (in this case, the solvent) on the electrostatic force, which is represented by the dielectric constant ε.

Electrochemistry Formula Templates – Kohlrausch Law and its Application
Electrochemistry – Kohlrausch Law
The total of the ionic conductivities of cations and anions, each multiplied by the number of ions present in one formula unit of the electrolyte, is the molar conductivity of an electrolyte at infinite dilution. Λm0 is the limiting molar conductivity. Λ∞cation is used for the molar conductivity of cation. Λ∞cation is used for molar conductivity of anion.

Electrochemistry – Molar Conductivity
In the calculation of molar conductivity at infinite dilution for a weak electrolyte. Here Λ use for molar conductivity.

Electrochemistry – Degree of Dissociation
The Kohlrausch law is used for the calculation of the degree of dissociation. α is used to denote degree of dissociation. Ka is a dissociation constant.



Electrochemistry – Sparingly Soluble Salt
In the calculation of solubility of a sparingly soluble salt. As the solution is saturated but infinitely dilute. Ionic mobilities at infinite dilution are denoted by the letter ‘u. The study of chemical reactions that cause electrons to move is known as electro-chemistry. This flow of electrons is known as electricity, and it can be generated by electrons moving from one element to another in an oxidation-reduction reaction. Ionic mobilities at infinite dilution are denoted by the letter ‘u.’κ shows conductivity of salt,Λ is for molar conductivity.
Molarity = solubility

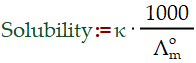
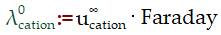

Electrochemistry Formula Templates – Electrochemical Cell Potential
Oxidation potential refers to an electrode’s tendency to lose electrons, while reduction potential refers to its tendency to absorb electrons. The EMF of a due cell is a measure of the free energy change that determines the total reaction’s chance of occurring. The reaction takes place, if EMF becomes positive.
E0Cell::=[Standard reduction potential of cathode E0cathode] – [Standard oxidation potential of anode E0anode].

Electrochemistry Formula Templates – Nernst Equation
When the conditions are not standard (concentrations not 1 molar and/or pressures not 1 atmosphere), the cell potential must be determined. Ecell potential at non-standard state condition, Ecell0 standard state cell potential, R constant (8.31 J/mole K),T absolute temperature (Kelvin scale), F Faraday’s constant (96,485 C/mole e-), n number of moles of electrons transferred in the balanced equation for the reaction occurring in the cell Q reaction quotient for the reaction and Mn+ active mass of the ions. For simplicity, it may be taken as equal to the molar concentration of the salt.
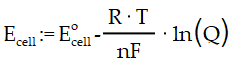


If the temperature of the cell remains at 25oC, the equation simplifies to

For a reduction reaction, use the Nernst equation for a single electrode reduction potential.
Mn++ ne– → nM


Electrochemistry Formula Templates – Gibbs Free Energy Change
In an electrochemical reaction, the Gibbs free energy change can be expressed as the potential difference. n is the number of electrons transferred in the reaction (from balanced reaction), F is the Faraday constant (96,500 C/mol) and E is potential difference/EMF of cell.


Electrochemistry Formula Templates – Concentration Cell
Concentration cells are electrochemical cells in which the emf is generated by material transfer from one electrode to the other due to a concentration difference between the two electrodes.
For concentration cells in general:

Electrochemistry Formula Templates – Equilibrium Constant
The free energy change in the form of equation can be related to the equilibrium constant (K), assuming unit activity for electrons.

Electrochemistry Formula Templates – Mass-Transfer limited current
Where represents the concentration of the electrochemically active species that gets converted at the electrode, m represents mass transfer coefficient, A is area that involve in transfer of electron. The expression for the mass-transfer limited current il [A] is given b;

Electrochemistry Formula Templates – De-Bay Huckle Theory Formula
In order to calculate the activity ac of an ion C in a solution, firstly know about the concentration (C) and the activity coefficient (γ), Cθ is the concentration of the chosen standard state, e.g. 1 mol/kg if molality is used.γ is the activity coefficient of C. C is a measure of the concentration of C.

The De-bay Huckle limiting law can be used to calculate an ion’s activity coefficient in a dilute solution with a known ionic strength.

z is the charge number of ion species i,q is the elementary charge,k is the inverse of the De-bay screening length,ε0is the permittivity of free space,εr is the relative permittivity of the solvent,,kΒ is the Boltzmann constant,NA is the Avogadro constant,I is the ionic strength of the solution,A is a constant that depends on temperature, T is a constant that depends on temperature.

Electrochemistry Formula Templates – Hydrogen Electrodes
In electro chemistry, the Normal Hydrogen Electrode (NHE) or Standard Hydrogen Electrode (SHE) is the standard reference point for standard electrochemical reduction potentials, with its potential, E°, assigned as 0.0000 volts (V) at all temperatures by convention.


Electrochemistry Formula Templates – Saturated Calomel Electrode
The potential of the mercury(I) chloride RE is defined by the chloride concentration in the filling solution, and can be calculated as shown below.

Electrochemistry Formula Templates – Cottrell Equation
In electro chemistry, the Cottrell equation is derived from Fick’s second law of diffusion and predicts the variation of the current in time, when a potential step is applied under conditions of large over potential where D0 [cm2 sec-1] is the diffusion coefficient for the species O, and A [cm2] is the electrode area.

Electrochemistry Formula Templates – Reference Electrodes
Electrochemistry – Mercury Sulfate Reference Electrode:
The reference potential of the MSRE is calculated as follows:
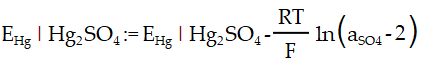
Electrochemistry – Mercury(II) oxide reference electrode:
The mercury(II) oxide (mercuric oxide) RE is limited to use in highly alkaline solutions due to the stability of the oxide. The potential of the mercury(II) oxide RE is independent of the electrolyte, but depends on the activities of both OH and H2O.

Electrochemistry – Silver/silver chloride reference electrode (Ag|AgCl):
The silver–silver chloride RE is the most regularly used RE due to its simplicity, inexpensive design, and nontoxic components. The potential is calculated as follows:

Silver/silver cation reference electrode (Ag|Ag+):
This RE is typically used as a non-aqueous RE with AgNO3 and a supporting electrolyte dissolved in MeCN or other organic solvents as the filling solution. where is the degree of ionization of Ag in the solvent, the molality of Ag+, and the molal ionic activity coefficient of Ag+ in the solvent The potential can be calculated as follows:
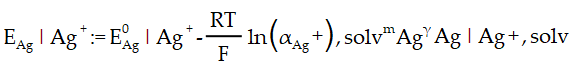
Electrochemistry Formula Templates – Ultramicroelectrode (UME) Response time and Resistance
In seeking to make transient measurements, the electrochemical cell must respond to the applied potential waveform much more rapidly than the process one is seeking to measure. Where R is the cell resistance (), C is the total inter facial capacitance (F), r0 is the electrode radius (cm) and is the conductivity of the solution (S cm1)

For an electrode immersed in a dilute solution of supporting electrolyte that does not contain any electro active species, a charging current, ic, will flow following a potential step according to equation:

The solution resistance for a disk-shaped UME is inversely proportional to the electrode radius. R increases as the electrode radius decreases.

Electrochemistry Formula Templates – Double Layer Capacitance of UME
This process causes electrons to flow into or out of the surface giving rise to the charging or capacitive response. The double-layer capacitance for a disk-shaped UME is proportional to the area (A = πr02) for an inlaid disk UME) of the electrode surface. Where Cd is the specific double-layer capacitance of the electrode.

Electrochemistry Formula Templates – Ion Selective Electrode Potential (ISEs)
Where zI is the charge of the analyte ion, I, aIw is its activity in the sample solution, and the constant term, EI0, is unique for the analyte

Electrochemistry Formula Templates – Nikolsky Eisenman equation and phase boundary potential
The influence of the interfering ions on the potentiometer responses has been described using the Nikolsky–Eisenman equation. where KIJpot is the selectivity coefficient for the analyte ion, I, against an interfering co-ion, J, and aJ and zJ are the sample activity and charge of the interfering ion.

Electrochemistry Formula Templates – Selectivity Coefficient
The selectivity coefficient can also be determined by measuring the cell potentials at different analyte concentrations in the presence of a fixed concentration of an interfering ion, where a I(DL) is the analyte activity at the detection limit and aJ(FIM) is the fixed activity of the interfering ion.

Electrochemistry Formula Templates – Potentiometric Response in the Mixed Ion Solution
The selectivity coefficient, however, can still be used in the new model to quantify the Potentiometric responses in the mixed ion solution. For example, the Potentiometric responses to a monovalent cation in the presence of a divalent cation are given as:

Electrochemistry Formula Templates – Semiconductor Electrode
Band theory of solids:
The time-dependent Schrödinger equation for a free electron is written as


Electrochemistry Formula Templates – De Broglie Wavelength
Based on the de Broglie wavelength associated with microscopic particles, the linear momentum and kinetic energy of the electron in a single crystal can be expressed in terms of k by the expressions:


References
Carl H. Hamann, Andrew Hamnett, Wolf VielStich Electrochemistry 2007 Wiley-VCH
Samuel Glasstone An Introduction to Electrochemistry 2007 Wiley-VCH
Allen J.Bard Electrochemical 2e Student Sol. Manual: Fundamentals and Applications 2008 John Wiley & Sons
